In the last quarter of 2013, Xander*, a Filipino living with HIV, noted the “cutting of the (amount of the) antiretroviral medicine (ARV) supplies provided me, from the usual stocks that would last me over three months to only a month,” he recalled. At that time, “I didn’t even ask about the limited supply; like most, I just… assumed the people at my treatment hub knew what they’re doing.”
Xander also “took this as somewhat of a non-issue,” he admitted, “considering that I know of other people living with HIV (PLHIV) in other parts of the Philippines who receive even less supplies of ARVs from their treatment hubs. According to them, because of the lack of supplies of ARVs, there are those who have to borrow ARVs from other PLHIV, and there are those who end up not (complying with their antiretroviral therapy or ART). Following assurances (from people in my treatment hub) that the shortage is temporary, my issue seemed somewhat trivial.”
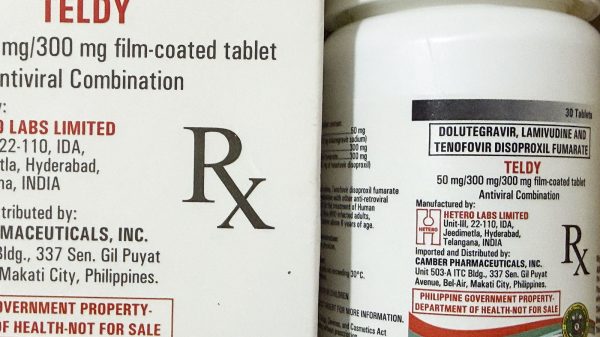
But already halfway through February 2014, the providing of limited supplies continues. And – in an earlier communiqué from blogger Pozzie Pinoy, who coordinated with Dr. Rossana Ditangco, research chief at the Research Institute for Tropical Medicine-AIDS Research Group (RITM-ARG, one of the treatment hubs in the country) – as of February 6, most Philippine HIV treatment hubs give out supplies for only either two weeks or one month. Particularly, there is shortage in the supplies of Lamivudine and Tenofovir; the rest of the ARVs are supposedly not affected.
According to Dr. Ditangco, the limited supply is “because of the delay in the delivery of (ARVs) to the Department of Health (DOH).”
Though Dr. Ditangco also added that “we are expecting the stocks to arrive within this month (February),” the likes of Xander have started to panic in the face of the shortage of ARV supplies. “That we’re running out of meds is the word on the streets,” he said.
TREATMENT IN PHL
Currently, Filipino PLHIV who are Philippine Health Insurance Corp. (PhilHealth) members or qualified dependents confirmed with HIV infection who undergo treatment in a DOH-accredited treatment hub are covered by the OHAT package. Coverage under this package includes treatment in accordance with the guidelines set by DOH on ART among adults and adolescents with HIV infection.
PhilHealth’s benefit package is, by the way, an add-on to Global Fund’s Transitional Funding Mechanism, which the country receives to support essential services, such as ART for eligible patients and outreach work among most-at-risk populations.
NO SHORTAGE, PER SE
According to Dr. Jose Gerard Belimac, head of DOH’s National AIDS/STI Prevention and Control Program, there is no delay in the procurement of ARVs, just as there is no “official pronouncement from the DOH to the treatment hubs to control (the distribution of ARVs) because of a delay in the procurement (of ARVs),” he said in an exclusive interview by Outrage Magazine. Belimac stressed that “for now, all the ARVs that we promised to provide to the patients are available.”

According to Dr. Jose Gerard Belimac, head of DOH’s National AIDS/STI Prevention and Control Program, there is no delay in the procurement of ARVs, and that “for now, all the ARVs that we promised to provide to the patients are available.”
However, “we acknowledge that it is also really very difficult to forecast the needs of the PLHIV because (they) change regimen from time to time for many reasons – for personal reasons, for clinical reasons,” Belimac said.
What happens, according to Belimac, is that DOH comes up with a forecast to respond to specific needs, but these needs change after the supplies are already acquired. As such, the giving out of ARVs to the patients does not always go as smooth as desired. Particularly, when the needs of one treatment hub are considered more urgent than the needs of other treatment hubs, “borrowing” of supplies happen.
Belimac nonetheless stressed that “this is something we are trying to resolve… to ensure the continuity of treatment for PLHIV.”
FUTURE FEAR
On February 19, Dr. Ditangco reported that the DOH will already deliver ARVs to the treatment hubs.
Reportedly, the Lamivudine-Tenofovir-Efavirenz stocks came in as a three-in-one tablet (1,200 mgs). The supplies of Lamivudine and Tenofovir as individual tablets remain critical level. And there are no known issues concerning the stocks of Lamividine-Zidovudine + Nevirapine combination.
However, another HIV-positive blogger – advocatehiv – listed down some of the developments in select treatment hubs he contacted. According to him, RITM-ARG, for instance, already confirmed that the ARVs have arrived from DOH, but it is still enforcing a one bottle issuance policy at the moment. Meanwhile, PLHIV from Philippine General Hospital (Sagip) reportedly already received SMS saying that the ARVs are already available, but they are to surrender all remaining Lamivudine and Tenofovir tablets. Outside Metro Manila, at the Corazon Locsin Regional Hospital (Bacolod-HACT), there reportedly remains shortage of Lamivudine and Tenofovir; they have yet to verify the arrival date of ARVs from Manila.
The differing practices of treatment hubs are dictated by who run them.
Sighing in relief, Xander still remains worried that the problem could surface again in the future. For Xander, “I don’t want to tell DOH and the treatment hubs how they should do their job, but if they pays attention to the continuing growing rate of HIV infections in the country, then they should recognize the need for it to be better prepared. Part of this preparedness is anticipating the ARV supplies that will be needed by Filipino PLHIVs. That way, the supplies will always be there.”
Because for Xander, in the end, “while the people who should deliver the services debate on whether there’s a problem or not, it is us – the people affected by the problem – who have to suffer.” (Article filed with John Ryan N. Mendoza)
*NAME CHANGED AS REQUESTED BY THE INTERVIEWEE