Took them long enough…
Xander (not his real name) has been HIV-positive since December 2012; and from the very first time he took his antiretroviral (ARV) medicines, “it has always been LTE (for lamivudine, tenofovir, efavirenz),” he said.
There have been adverse side effects – e.g. “At night, my dreams are so vivid I am unable to distinguish what’s real or what’s not,” Xander said, adding with sadness that “the doctor just told me to ‘Drink more water!’ as if that’d solve my problem.”
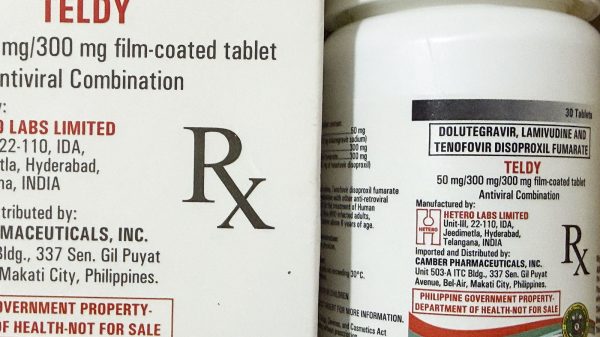
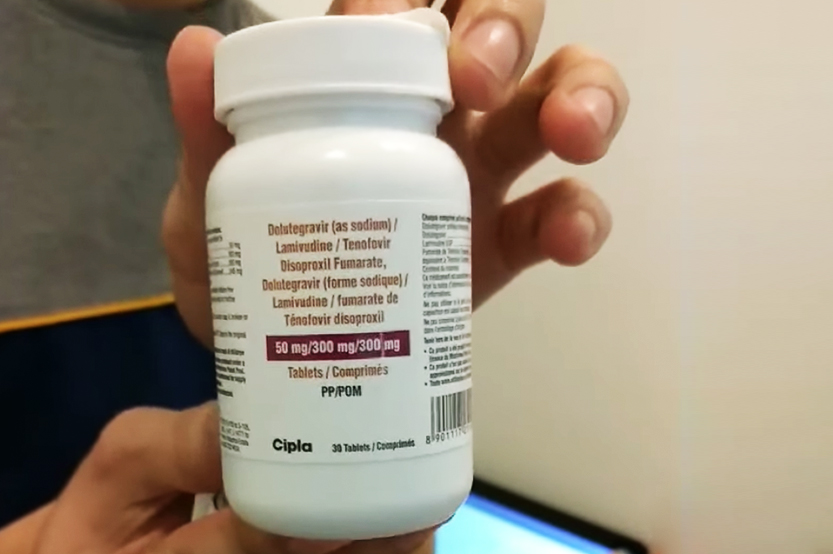
Those like Xander may now have their ARVs changed, with the Department of Health (DOH) introducing LTD (for lamivudine, tenofovir, dolutegravir) in the country.
This December, the Global Fund for treatment of people living with HIV (PLHIV) donated 197,260 bottles of LTD. The stock is being managed by the DOH’s Disease Prevention and Control Bureau – National HIV, AIDS and STI Prevention and Control Program.
The first tranche of 98,630 bottles were already allocated to various Centers for Health Development, while the last tranche (98,630 bottles) are expected to arrive in the country before the end of the year.
In an advisory signed by Undersecretary of Health Dr. Myrna Cabotaje, the DOH stated that based on the National Plan for LTD Introduction, the new ARV will be introduced from now until December 2021. These sub-populations of PLHIVs can benefit from LTD:
- ART-naive adults, adolescents and children (30 kg and above), excluding TB patients on rifampicin-based regimen
- PLHIVs on LTE with severe adverse events (dizziness, insomnia, abnormal dreams, anxiety, depression, mental confusion, convulsions, hepatoxicity, severe skin and hypersensitivity reactions and gynecomastia)
- Patients with treatment failure to zidovudine (AZT) and abacavir (ABC)-based regimens
Xander knows he has to broach his concerns to the doctor in his treatment hub again to ascertain if he can shift from LTE to LTD. And “the DOH really took its time,” he mused, adding that hopefully, “this is just one of the steps wherein Filipino PLHIVs actually already start being able to access life-saving HIV meds already widely available in more developed countries.”